About our research group/lab
Our research
Background
At Biomedical Engineering, for over 50 years now, engineers, scientists, clinicians, and companies team up to address the challenging task of diagnosis, treatment, and monitoring of cardiovascular diseases. The group, part of the Cardiology department of Erasmus MC, forms a strong multidisciplinary team to develop novel diagnostic and treatment strategies. The work covers the entire pathway from basic concepts and modelling to laboratory work, patient studies, and commercialization.
Overall aim
Imaging, modelling and data interpretation stand at the basis of disease diagnosis, treatment, therapy guidance and monitoring, such that clinicians can provide better prognosis for the patient. We aim to develop and study unprecedented techniques to image the cardiovascular system and improve treatment of the patient.
Research focus area
The body contains many dynamics, e.g. the heart motion and blood flow, but also biological interactions between cells and their environment. We study and predict these complex dynamics, through state-of-the-art (and beyond) high frame rate techniques, thus being able to show the phenomena in slow-motion. For this, we build and use volumetric transducers and systems for echography, camera systems for in vitro visualization, and advanced computer models to predict and analyze the physics and biology underlying the phenomena. We challenge ourselves to develop the imaging, understand and model the physics and biology, improve the signal processing, and interpret the results with clinicians to provide actual information needed for better treatment of patients.
Our four research themes
Our projects
4D-Flow Echocardiography for Diastolic Functional Imaging of the Left Ventricle
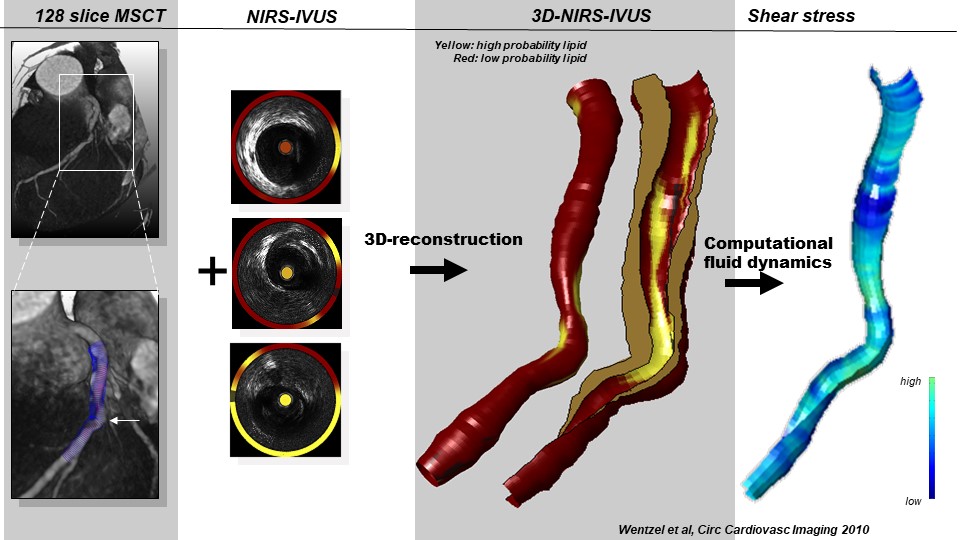
Biomechanics and Atherosclerosis
Biomechanics in Congenital Cardiology
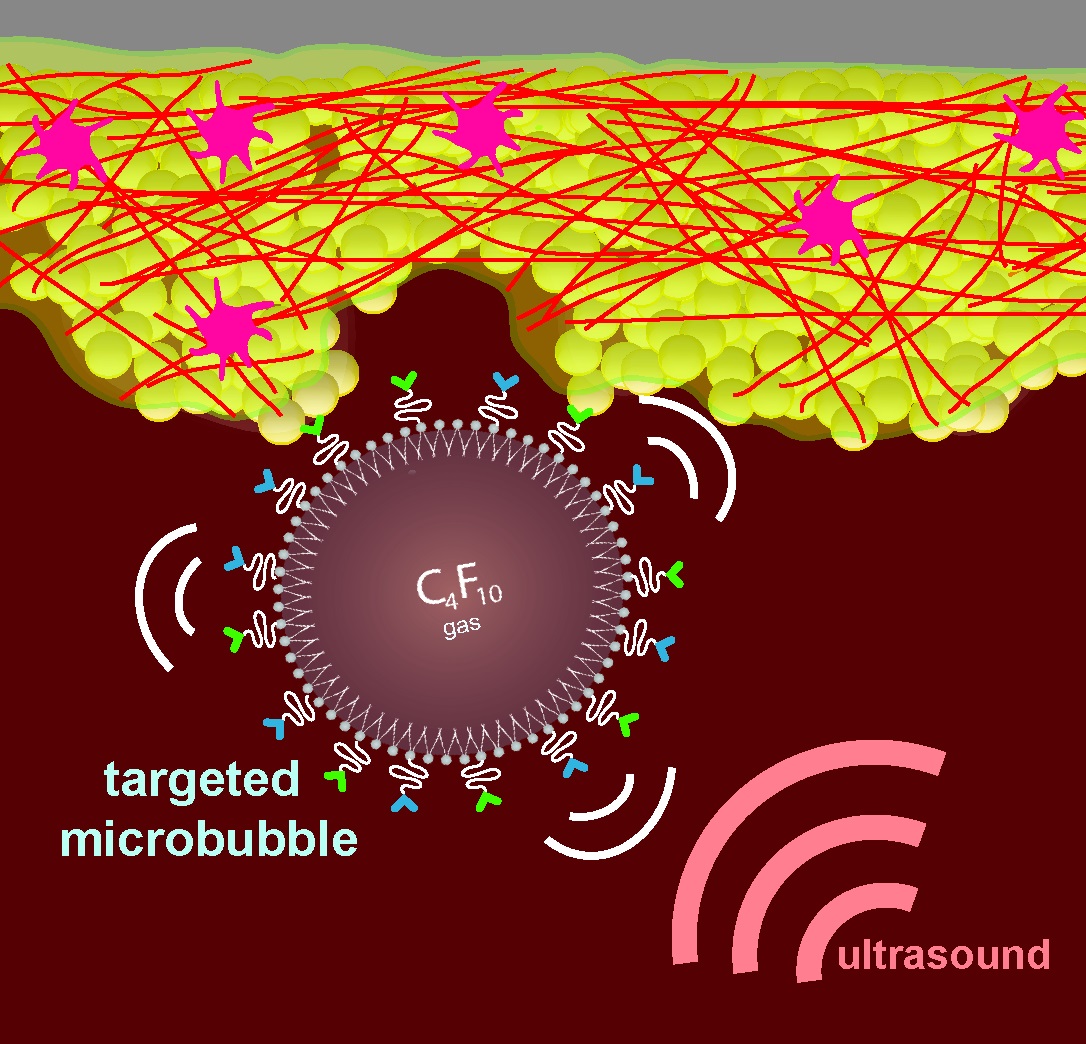
BUBBLE CURE
BUBBLE QUEST
Cardiac Mechanics
EFFECTS - Echocardiographic Tissue Stiffness measurements
Human disease model for atherosclerosis
Imaging atherosclerotic plaque lipidomics in vivo
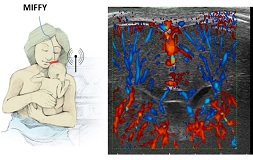
MIFFY- Monitoring infant brain perfusion by echography
Molecular imaging of atherosclerosis
PARFAIT
QUANTO-3D-myocardial-perfusion-imaging-with-echography
Rapid Innovation of Minimally Invasive Instruments
Seeing the heart attack (and more) before it happens
Theranostic-phospholipid-coated-ultrasound-contrast-agents
ULTRA-X-TREME X-Flow
Key Publications
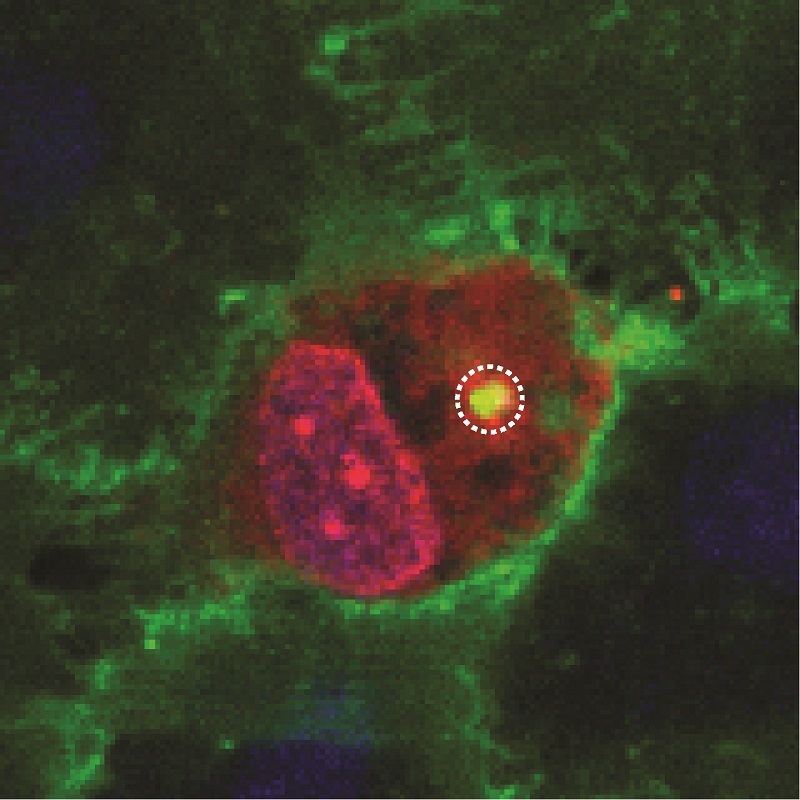
Meijlink et al (2024): Ultrasound-activated microbubbles mediate F-actin disruptions and endothelial gap formation during sonoperation
Voorneveld et al (2019): High-Frame-Rate Echo-Particle Image Velocimetry Can Measure the High-Velocity Diastolic Flow Patterns
Hoogendoorn et al (2019): Variation in Coronary Atherosclerosis Severity Related to a Distinct LDL (Low-Density Lipoprotein) Profile: Findings From a Familial Hypercholesterolemia Pig Model
Strachinaru et al (2019): Naturally Occurring Shear Waves in Healthy Volunteers and Hypertrophic Cardiomyopathy Patients
Visscher et al (2019): Data Processing Pipeline for Lipid Profiling of Carotid Atherosclerotic Plaque with Mass Spectrometry Imaging
Iskander-Ritz et al (2019): Photoacoustic imaging for guidance of interventions in cardiovascular medicine
Collaborations
Collaboration within Erasmus MC
- Cardiology
- Radiology
- Neuroscience
- Neonatology / Erasmus MC Sophia
- Erasmus MC Cancer institute
Collaborations outside Erasmus MC
We work within several local, national and international frameworks:
- Medical Delta
- Dutch Society of Medical Ultrasound
- Dutch Royal Academy of Science
- World federation for ultrasound in medicine and biology
- IEEE Ultrasonics, Ferroelectrics and Frequency Control Society
- International Contrast Ultrasound Society
Within Biomedical Engineering, we are organizing 3 international annual conferences:
- The International Symposium on Biomechanics in Vascular Biology and Cardiovascular Disease
- Optics in Cardiology
- The European symposium on Ultrasound Contrast Imaging and therapy
Funding & Grants
The group is well-funded with grants from many sources.
These include Open Technology projects and personal grants from the Dutch Research Council (NWO), public-private partnership grants such as the Medical Delta and a NWO/Dutch Heart Foundation grant, Open and Personal grants from the European Research Council, and many local initiatives such as the TU Delft/Erasmus MC Convergence, and the Erasmus MC fellowship. See specific pages per subgroup.
The group hosts a NWO PIONIER, A Simon Stevin Master, 2 ERC laureates, 3 Veni’s, 3 Vidi’s, 1 Vici and a marie curie fellow.
Career opportunities
Available Bachelor/Master projects
Project: Development and characterization of PVDF transducer for Photoacoustic imaging
Do you want to work on transducer development for photoacoustic imaging? Do you want to enhance your knowledge on medical imaging and the electronics behind transducer technology? Then this is an interesting project for you!
Aim
In this project, we aim to develop and characterize a Polyvinylidene Difluoride (PVDF) based piezoelectric transducer array to enhance sensitivity and bandwidth for receiving photoacoustic signals.

Why it is important
Photoacoustics is an imaging modality that uses light illumination in combination with ultrasound detection to image different molecules within the human body. These signals are broadband in nature due to the underlying physics. In addition, in vivo targets such as the atherosclerotic plaques in the carotid artery are deep-seated. They generate very weak PA signals in comparison to conventional ultrasound signals. Therefore, there is a strong need to develop transducers that are better suited for photoacoustic signals.
What will you learn
You will work closely with a technician and a postdoctoral researcher in building the electronics for the PVDF transducer. You will also be involved in performing experiments to characterize the transducer’s sensitivity and response. You will gain experience in working with laser system, optical components and data acquisition with the Verasonics research ultrasound scanner.
Requirements and expectations
You have some basic experience with building circuits and designing amplifiers. You know the fundamentals of hydrophones, transducers and piezoelectric properties. You have used basic lab equipment such as the oscilloscope, function generator, pulser/receiver, etc. Experience with using hydrophones for performing transducer characterization would be desirable. You are eager to learn, try and perform experiments.
Contact
Note: The project has to be performed at ErasmusMC.
Project: Analysis of 3D right ventricular shape from echocardiography
Introduction
The functioning of our hearts is of vital importance to physical wellbeing. The right half of the heart is responsible for collecting the returning blood from the main circulation and pumping it to the lungs, where it is resupplied with oxygen. When the functioning of the right ventricle is compromised, venous pressure will rise and lung function will be decreased, leading to severe health problems on the long term.
The right ventricle has a complex 3D shape, similar to a thick triangle that is wrapped around the ellipsoidal left ventricle. It consists of an inlet zone, a main (apical) part and an outflow tract. It contracts in a complex peristaltic manner. For diagnosis of right ventricular function, 3D cardiac ultrasound can be used. We have collected about 200 4D (3D+time) echocardiography studies of the right ventricle in various congenital heart disease patient groups as well as healthy controls. These studies have already been semiautomatically analysed using Tomtec 4DRV software. We would like to perform further analyses on this large collection of 3D+T geometrical shapes of the right ventricle, such as local curvature, different decompositions of shape change, right ventricular global motion and local deformation. We want to relate those to patient characteristics. MRI analyses are available on part of the patients as well.
Purpose of this project
Implementation of several shape analyses of the right ventricle. Basic code for reading the shape files and original image files and visualizing them is available. Depending on the skills and technical and clinical interest of the candidate, the assignment can be limited or extended to include more complex spatiotemporal shape analyses, statistical analyses of the results, visualization/comparison tools etc.
Short outline of the project (6-9 months)
- Literature study on existing RV shape analyses
- Design, implementation and testing of shape analysis methods
- Generation of results on total data set
- Optional: writing of publication, implementation of additional methods
Requirements
- Bachelor or master students Biomedical Engineering, Informatics, Clinical Technology or similar
- Interest in medical imaging, biomedical research
- Programming experience in MatLab, Python or C
For more information, please contact:
Project: Volumetric photoacoustic imaging of kidney
Introduction
Photoacoustics (PA) is a rapidly developing imaging modality which provides information about the molecular composition of biological tissue. The photoacoustic signal is generated by so-called chromophores, such as biological molecules that absorb light, which can transform absorbed light into an ultrasound signal. The presence of certain types of chromophores can be a valuable marker indicating the condition of biological tissue. The accurate quantification of certain types of chromophores can improve clinical diagnosis.
However, at each moment, only a limited area of human tissue can be exposed by a light (laser) which impedes volumetric analysis of present chromophores. The goal of this project is to quantify present chromophores in large organs, e. g. kidney, which are normally challenging to be fully imaged with PA.
Clinical problem description
The main application of this project is kidney evaluation before transplantation . Only functioning and free of injuries kidney are suitable for transplantation. First, the kidney will be inspected for presence of injuries like necrosis or fibrosis. Second, the functioning of the kidney will be evaluated by measuring blood perfusion through the kidney’s cortex.
Technical problem description
We are using the pre-clinical Vevo LAZR-X photoacoustic system (Fujifilm Visual Sonics, Toronto, Canada). It comprises a laser source in a combination with high-frequency US probe. The probe is mounted in a translational stage facilitating 3D acquisition which volume is much smaller than the volume of the kidney.
Your task would be to develop a method for full volumetric reconstruction of the kidney based on small acquired sub-volumes.
You will:
- Get experience in PA and US imaging
- Principles of PA imaging
- Basics of B-mode US
- PA spectroscopy
- Solve a clinically relevant problem
Requirements
- Confident user of Matlab or Python
- Understanding of medical imaging
- Being ready to read, write and code =)
For more information, please contact:
Project: The effect of fast microbubble motion on contrast pulse sequences
Introduction
Ultrasound contrast agent (UCA), most commonly in the form of 1-10 µm microbubbles consisting of a lipid-monolayer shell with an inert gas core, are used to opacify the blood pool in ultrasound imaging. A more recent application of UCA is as tracer agents for echo-particle image velocimetry (echoPIV) where high frame-rate ultrasound imaging is used to track the motion of the microbubbles as they flow with the blood.
In order to isolate the microbubble signals from unwanted surrounding clutter signal (e.g. from surrounding tissue, valves and/or the lungs) contrast pulse sequences, which take advantage of the non-linear microbubble response to the phase and magnitude of the ultrasound pulse (in contrast to the linear scattering from tissue).The most common sequences used are sequentially transmitted pulses of alternating polarity (pulse inversion) or amplitude (amplitude modulation), which cancel the linear components of the scattered signal while preserving the non-linear components.
However, these contrast pulse sequences assume that there has not been significant microbubble motion over the duration of the received pulses, and that any phase changes in the received pulse are due to the microbubble response and not its motion. Simplistic simulations suggest that this is not the case for fast blood flows (0.6 – 1.2 m/s) which are common during filling and ejection in the left ventricle. The fast blood flow interferes with the cancellation of the linear component and may even cancel the non-linear components at certain combinations of bubble velocity and imaging frame-rate. These findings are also suggested from in-vivo and in-vitro experimental data. This is a serious problem in applications such as echo-particle image velocimetry, where the microbubbles need to be tracked between frames to obtain the velocity field information.
The purpose of this study is to investigate the effect of inter-pulse bubble displacement on the efficacy of contrast pulse sequences and determine how best to compensate-for or avoid this issue.
Short outline of the project (6-9 months)
- Literature study on ultrasound imaging and ultrasound contrast agents.
- Perform realistic bubble response simulations, using a Rayleigh-Plesset model (or variation thereof) and obtain the acoustic response of bubbles to each pulse in the pulse sequences.
- Determine how different contrast pulse sequences are affected by fast microbubble motion.\
- Corroborate the simulation results with in-vitro experimental data.
- Investigate the possibility compensating for the bubble motion between pulses.
- Write-up of final report.
Requirements
- Background in biomedical engineering, physics or similar
- Knowledge of imaging processing and signal processing techniques
- Programming experience in Matlab and/or Python
For more information, please contact:
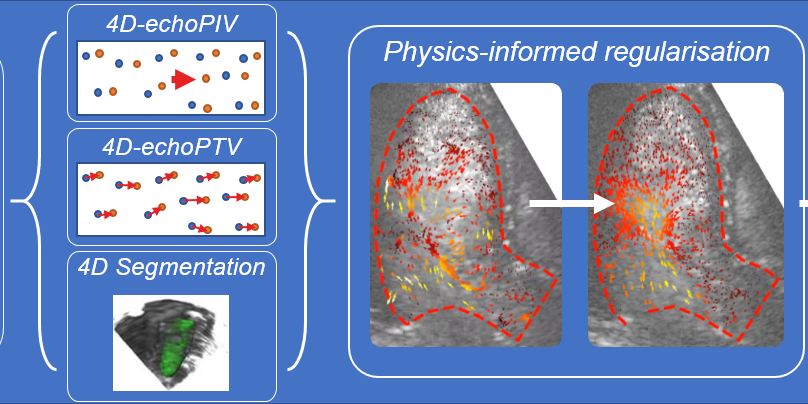
Project: Conditioning Navier-Stokes based velocity field regularization with sparse high-fidelity data
Introduction
Vector flow imaging (VFI), a relatively recent development in diagnostic ultrasound research, describes a group of flow imaging methods that quantify both the magnitude and angle of the blood velocity vectors in the organ of interest (e.g. the heart). This is in contrast to traditional Doppler ultrasound which only estimates the velocity component on the axis towards the transducer.
Echo-particle image velocimetry (echoPIV) is a VFI method that tracks the motion of intravenously injected microbubbles (ultrasound contrast agent) over time to estimate the blood velocity field. We have shown previously that echoPIV can accurately estimate the blood velocities in the heart (Voorneveld et al., 2019). However, the resulting velocity fields are often corrupted by noise and/or dropouts, thus requiring regularization. Regularization of velocity fields is classically done with naïve Gaussian smoothing kernels which tend to over-smooth jet-like flow structures, resulting in underestimation of the true velocity magnitudes.
A novel approach to regularization of these time-resolved velocity fields is to fit the data with physically informed models (Grønli et al., 2018), such as the Navier-Stokes and continuity equations. However, when using these models we have noticed that the high velocity flows during ventricular filling and ejection are over-dampened (Figure 1). A work around could be to insert high fidelity data from the high velocity regions into the fitting process, so that the final solution preserves these regions. This can be achieved with pulsed-wave doppler estimates or by using echo-particle tracking velocimetry (which tracks a sparse distribution of individual microbubbles).
The objective of this study is to develop and assess the efficacy of supplementing physics informed regularization models with sparsely located high fidelity velocity data. The desired outcome is a smooth velocity field without over-damping of high velocity flow structures.
Short outline of the project (6-9 months)
- Literature study on particle image velocimetry, particle tracking velocimetry and fluid dynamics.
- Develop pipeline for integrating pulse-waved doppler / particle tracking velocimetry sparse velocity estimates as input to regularization model.
- Develop an in vitro or in silico experimental setup to assess performance of supplementing sparse velocity estimates.
- (Optional) Assess performance with in vivo left ventricular datasets.
- Write-up of final report.
Requirements
- Background in biomedical engineering, physics or similar.
- Knowledge of imaging processing and signal processing techniques.
- Programming experience in Matlab and Python. Optional: knowledge of tensorflow API.
For more information, please contact:
Our team
Principal Investigators
-

Prof. dr. ir A.F.W. (Ton) van der Steen, PhD
Head of Biomedical Engineering
-

Prof. dr. G. (Gijs) van Soest, PhD
Head of Invasive Imaging
-

Prof. dr. ir N. (Nico) de Jong, PhD
Professor Biomedical Engineering
-

Dr.ir. J.G. (Hans) Bosch, PhD
Associate Professor
-

Dr. ir. F.J.H. (Frank) Gijsen, PhD
Associate Professor
-

Dr. K. (Kim) van der Heiden, PhD
Associate professor
-

Dr. K. (Klazina) Kooiman, PhD
Associate professor, Head of Therapeutic Ultrasound Contrast Agents
-
Dr. ir. H.J. (Rik) Vos, PhD
Associate Professor
-

Dr. ir. J.J. (Jolanda) Wentzel, PhD
Associate professor, Head of Biomechanics Laboratory
-

Dr.ir. A.C. (Ali) Akyildiz, PhD
Assistant professor
-

F. (Francis) Kalloor Joseph, PhD
Assistant Professor
-

Dr. K.R. (Kirby) Lattwein, PhD
Assistant professor
-

Dr. ir. J.D. (Jason) Voorneveld, PhD
Assistant Professor
Postdocs
-

Dr. ir. A.F.M. (Annette) Caenen, PhD
-

Dr. A. (Anton) Nikolaev, PhD
Postdoctoral researcher
-

Dr. S. (Sowmiya) Chandramoorthi
Postdoctoral researcher
Phd Students
-

MSc. M. (Mervenur) Akkus
PhD candidate
-

MSc. B.P. (Brian) Berghout, MD
PhD candidate
-

Msc. L. (luca) Bontempi
PhD candidate
-

MSc. J.M.H. (Janneke) Cruts
PhD candidate
-

MSc. A.A.S. (Aaron) Doug Deen
PhD candidate
-

Msc. S. (Silke) Dreesen
PhD candidate
-

MSc. Y. (Yichuang) Han
PhD candidate
-

MSc. Y. (Yuyang) Hu
PhD candidate
-

I.L. (Imke) Fase-Jansen, MSc, PhD
Postdoc
-

MSc. H. (Hongchen) Li
PhD candidate
-

MSc. A. (Antonio) López Marín
PhD candidate
-

MSc. K.B.A. (Bram) Meijlink
PhD candidate
-

MSc. S. (Sander) Spiekhout
PhD candidate
-

Ir. A. (Katerina) Tziotziou
PhD candidate
-

MSc. G. (Geraldi) Wahyulaksana
PhD candidate
-

MSc. Y (Yuchen) Wang
PhD candidate
-

MSc. L.X. (Lucy) Wei
PhD candidate